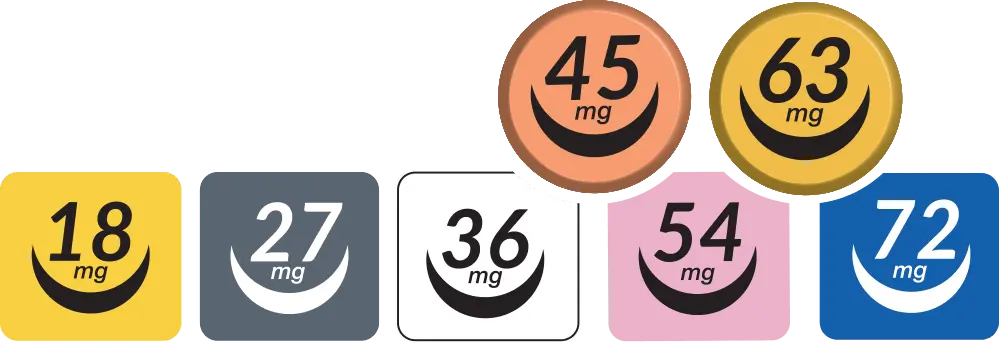
RELEXXII solves the dose gap
with 45 mg and 63 mg strengths

Finding the right dose in ADHD treatment can be
easier with smaller, 9 mg adjustments.1,2
Why choose RELEXXII?

-
Only branded
methylphenidate HCl to
offer 45 mg and 63 mg
strengths -
Unique Osmodex
Technology releases
RELEXXII at a
controlled rate -
Symptom relief within
an hour and maintained
throughout the day -
Savings offers to help
reduce out-of-pocket
costs
What is RELEXXII?

RELEXXII (methylphenidate HCl extended-release tablets) is a central nervous system (CNS) stimulant indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in adults (up to the age of 65 years) and pediatric patients 6 years of age and older.1
ADHD symptom control
that lasts all day

RELEXXII is an extended-release methylphenidate medication that uses unique Osmodex Technology. Osmotic pressure
gradually delivers medication throughout the day to help support symptom control from morning through early evening.
Flexible dosing to solve the dose gap

Finding the lowest effective dose of ADHD medication often requires careful adjustments to help balance symptom control
and tolerability. Some methylphenidate medications are only available in limited strength increments.

RELEXXII offers a dosing range with 9 mg step adjustments, including 45 mg and 63 mg strengths, enabling healthcare
providers to fine-tune treatment plans to keep patients on their lowest effective dose.

Support for access and affordability

Those starting or continuing ADHD medication may face access challenges with cost considerations or pharmacy
availability. RELEXXII provides resources to help support you.

Pay as little as $0 per month*
RELEXXII’s savings resources help eligible patients
access their prescribed treatment for less.
References: 1. RELEXXII package insert. Vertical Pharmaceuticals, LLC. 2. Childress AC, et al. Clin Pharmacol Drug Dev. 2025;0(0):1-7.
*Co-pay assistance is applicable to prescription coverage for eligible commercially insured patients. Offer void where prohibited. No income restrictions apply. Other restrictions may apply. Offer effective 04/2024. Alora Pharmaceuticals, LLC reserves the right to rescind, revoke, or amend this offer without notice. For full terms and conditions, click here.
All individuals depicted are models used for illustrative purposes only.







